|
8/18/2023 0 Comments Hydrogen bonding
Furthermore, \(H_2O\) has a smaller molar mass than \(HF\), but partakes in more hydrogen bonds per molecule, so its boiling point is consequently higher. This is because \(H_2O\), \(HF\), and \(NH_3\) all exhibit hydrogen bonding, whereas the others do not. We see that \(H_2O\), \(HF\), and \(NH_3\) each have higher boiling points than the same compound formed between hydrogen and the next element moving down its respective group, indicating that the former have greater intermolecular forces. However, when we consider the table below, we see that this is not always the case. Larger molecules have more space for electron distribution and thus more possibilities for an instantaneous dipole moment. 
This, without taking hydrogen bonds into account, is due to greater dispersion forces. When we consider the boiling points of molecules, we usually expect molecules with larger molar masses to have higher normal boiling points than molecules with smaller molar masses. For example, intermolecular hydrogen bonds can occur between \(NH_3\) molecules alone, between \(H_2O\) molecules alone, or between \(NH_3\) and \(H_2O\) molecules. They can occur between any number of like or unlike molecules as long as hydrogen donors and acceptors are present an in positions in which they can interact. Intermolecular hydrogen bonds: Intermolecular hydrogen bonds occur between separate molecules in a substance.For example, intramolecular hydrogen bonding occurs in ethylene glycol (\(C_2H_4(OH)_2\)) between its two hydroxyl groups due to the molecular geometry. In order for this to happen, both a hydrogen donor an acceptor must be present within one molecule, and they must be within close proximity of each other in the molecule. This occurs when two functional groups of a molecule can form hydrogen bonds with each other. Intramolecular hydrogen bonds: Intramolecular hydrogen bonds are those which occur within one single molecule.Hydrogen bonds can occur within one single molecule, between two like molecules, or between two unlike molecules. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'hydrogen bond.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Emily Underwood, Smithsonian Magazine, 6 Jan. 2020 When water molecules come up against the abrupt transition to air, many can no longer find partners for all their hydrogen bonds. Jennifer Ouellette, Ars Technica, 31 Mar. Meghan Herbst, Wired, The two main components of this body fluid are water and urea, a molecule that allows the hydrogen bonds to be broken and therefore reduces the viscosities of many aqueous mixtures. Quanta Magazine, 12 July 2021 Perhaps the most basic-and astonishing- of these concepts is the hydrogen bond, which holds together the literal stuff of life: water. Haseltine, Forbes, But that modest change allows Z to form a triple hydrogen bond with T, which is more stable than the double bond that holds together A-T. 2023 Additionally, five mutations in the central helical region, N764K, D796Y, N856K, L981F, and N969K introduce and facilitate additional hydrogen bond and hydrophobic interactions between the S2 trimers (Figure 4). 2019 The molecule has to have certain properties, like a number of hydrogen bond donors and acceptors. 2023 The hydrogen bonds with the oxygen to create water vapor, which is far easier to remove than atmospheric oxygen. 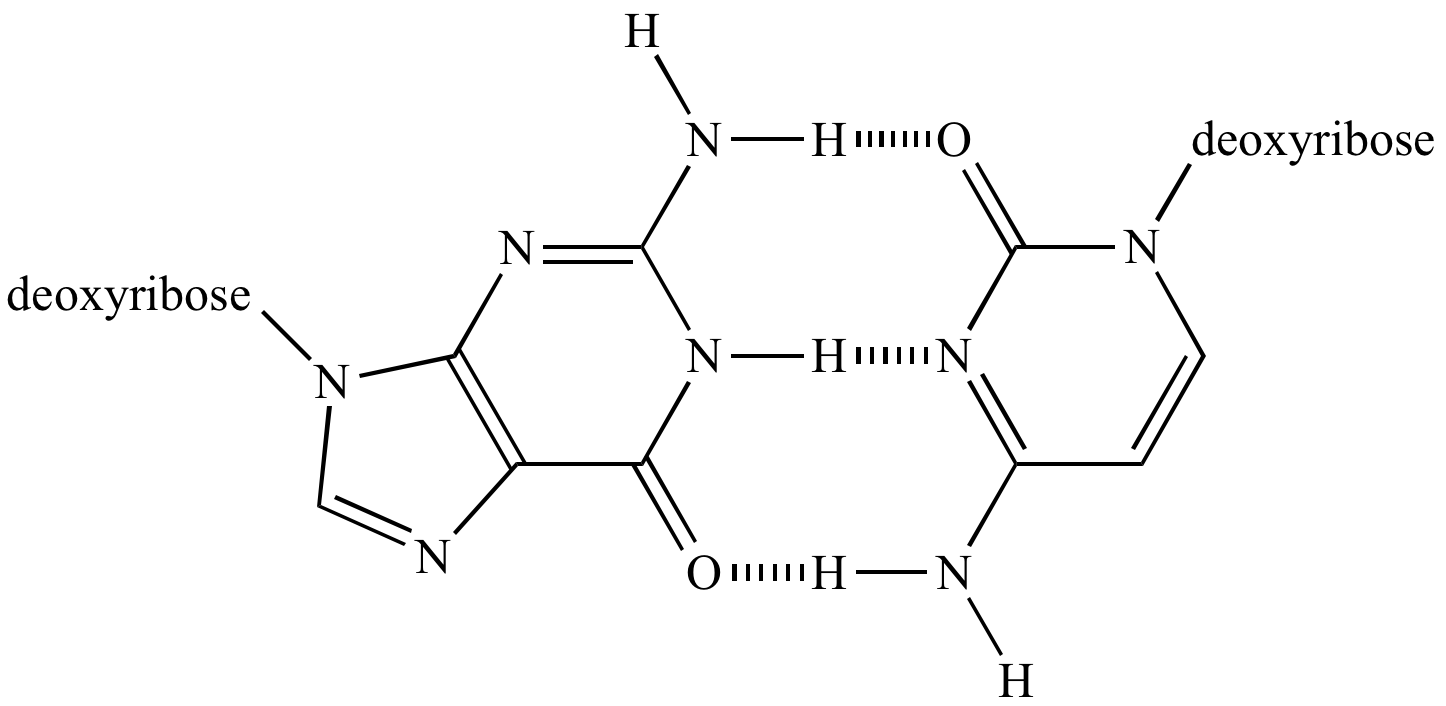
Recent Examples on the Web Water and salts create a hydrate, a rigid icy lattice supported by hydrogen bonds, in cold temperatures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
